It may not be surprising to learn that cancer is the top cause of death in the world. Yet, hearing that liver cancer is the second most common cause of death here in Singapore might make quite a few people respond in shock. The truth is, it causes almost 700,000 deaths globally each year!
Now, those of us who go out for bar nights will surely have the following questions running through our minds: What is Singapore doing about it? What causes liver cancer? Is there a cure for it?
We got some of our questions answered by Christy Man, Chief Strategy Officer of SCG Cell Therapy (SCG), a leading biotechnology company focused on the development of novel immunotherapies for infections and their associated cancers.
Q: How do liver cancers form?
Cancers typically start from a viral infection. The leading cause of liver cancer is particularly the Hepatitis B virus (HBV) which attacks the liver and can cause chronic diseases. Here’s a not-so-fun fact for us in Singapore (sorry!): HBV infection is the major cause of primary liver cancer (cancer that starts in the liver) in more than 80% of the Asian population. Primary liver cancer is also called hepatocellular carcinoma (HCC).
Q: Are there any cures at all?
Well, the good news is that there are a few therapies and treatments for liver cancer on the market like cell therapy, antibodies, and vaccinations against cancer. Right now, SCG is one of the only two companies in Singapore focusing on cell therapy – TCR-T Cell Therapy (SCG101) – to treat liver cancer. It is a form of immunotherapy, which is a form of treatment that uses the power of the body’s immune system to prevent, control, and eliminate diseases. Thankfully, it has led to major treatment breakthroughs for several diseases and cancers thus far!
Q: Wait, what’s ‘T-cell therapy’ and how does it compare to other cancer treatments for liver cancer?
T-cell therapy is a therapy which uses our body’s cells to fight cancer. Our bodies have circulating immune cells called T-cells that carry their proteins called T-cell receptors (TCR), which detect foreign antigens. Once T-cells receptors bind to foreign antigens, it will trigger signalling cascades that activate the immune system, to eliminate the foreign substance. However, with cancers, our T-cells are often unable to detect these foreign cancerous antigens, hence there’s a need for T-cells redirection and T-cell receptor reconfiguration.
By modifying T-cells through genetic engineering in a laboratory, and reinjecting them back into the patient’s body, the engineered T-cells can recognise and target cancer cell antigens better than before, enhancing the T cell fighting power to attack cancer cells.
While there are multiple treatments like target therapies or stem cell transplants available to treat other forms of cancers, as cancers vary depending on the tissue of origin, what may work for one cancer type may not be as effective for others.
Right now, there are limited available treatments in the market for patients who are in the advanced stages of cancer. What’s more, such treatments only guarantee a 3% survival rate in a 5-year relative period.
SCG’s SCG101 TCR T cell therapy addresses both the antitumor immune response, and promises antiviral activity to eradicate HBV (Hepatitis B Virus) infection – one of the primary drivers of liver cancer in Asia. In 66% of patients, improved tumour control and lesion shrinkage were observed.
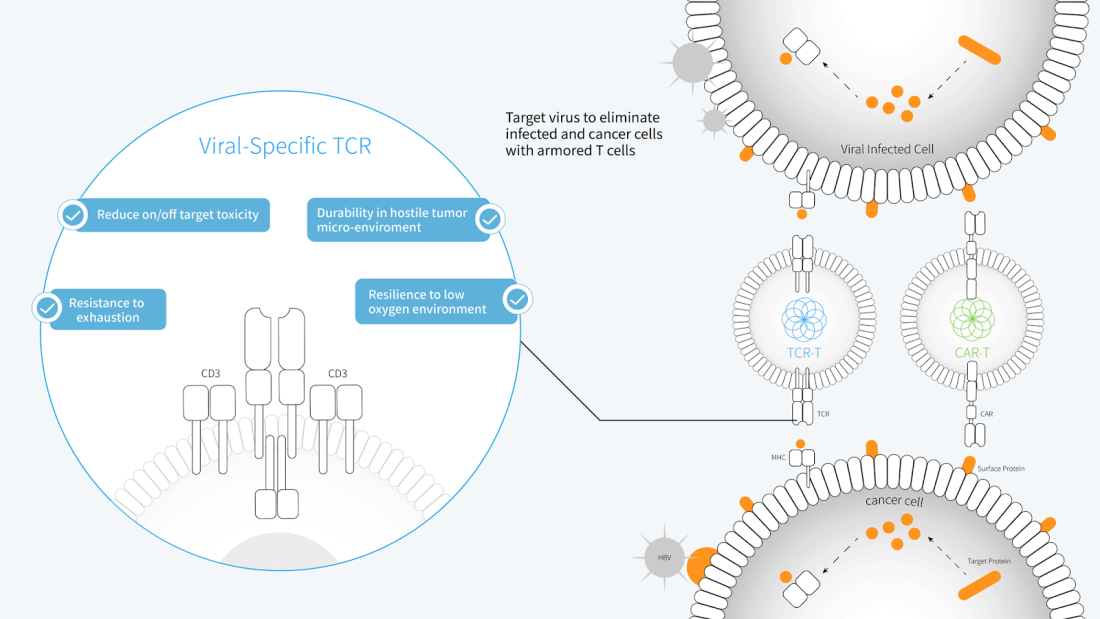 SCG’s CAR-T and TCR-T Cell Therapy Platforms
SCG’s CAR-T and TCR-T Cell Therapy Platforms
Q: So, what’s so great about SCG’s SCG101?
SCG101 is the first-in-the-world cell therapy treatment that has approvals to do multi-country clinical trials in China and Singapore. SCG101 is a TCR-T cell therapy targeting specific HBV cells involving cells and tissues derived from the same individual’s body. An HBV infection can lead to damage to the integrated hepatocyte genome (often known as chief liver cells) and leads to the occurrence of HCC (liver cancer!). SCG101 helps to redirect T cells to target HBV epitopes expressed on the cell surface and become a new therapeutic strategy for HBV-associated HCC. It’s like sending in a large army of ants that have been instructed to only head towards a single jar of sugar in a path without being distracted by other foods around!
Q: What, sounds too good to be true! Is there proof this treatment even works?
SCG is delighted to share clinical proof of concept (proof that it works!) from their most recent clinic that shows tumour control with lesion shrinkage observed in two out of three patients (66%) with advanced HBV-related HCC. These early interim results are promising and advance the clinical trials to the next stage! (Read their latest release here)
Q: Are Singapore research bodies and hospitals supportive of this treatment?
Fortunately, yes! SCG Cell Therapy is collaborating with multiple local organisations such as Health Science Authority, A*STAR, National University Hospital Singapore, and National Cancer Centre Singapore for clinical trial approvals and projects to help further progress cancer immunotherapy studies. Future patients will get the opportunity to receive this treatment whenever it is fully rolled out in hospitals in time to come.
Q: What available T-cell therapy is on the market right now, and what works so far?
The number of companies researching a cure for liver cancer locally is few. Currently, Kymriah, a type of T-cell therapy, is available on the market. The Health Sciences Authority (HSA) has currently approved one type of chimeric antigen receptor T-cell therapy, under the pioneer cell, tissue, and gene therapy products regulatory framework. Particular types of blood cancers are being treated with Kymriah.
Q: What is the availability of cancer treatment and cell therapies with SCG Cell Therapy for current patients?
Currently, phase I/II trials with SCG101 for HBV-related liver cancer are underway, with multi-country clinical trials in the U.S., China, and Singapore having been approved.
SCG101 clinical trials are ongoing at the National University Hospital (NUH) and the National Cancer Centre Singapore (NCCS). The clinical trials are also now open for patient recruitment – current patients can sign up for the trials to participate in cell therapy for cancer treatment.
SCG101 cell therapy clinical trials will also be conducted in other Southeast Asian countries, where the incidence of HBV-related HCC (HCC is the most common type of primary liver cancer) is high.
Q: What are the plans for the future of SCG’s T-cell therapy and platforms?
Cellular therapy could be the future of cancer treatments, and can potentially open doors to more advanced treatments soon.
As part of the Research, Innovation and Enterprise 2020 plan (RIE 2020), Singapore announced that S$80 million will be invested to develop core capabilities required for cell therapy manufacturing. This includes strategic programmes that address the major gaps in advanced cellular therapies manufacturing. It aims to scale manufacturing, automation, cell therapy product development and new technologies for assessing the safety, efficacy and quality of cells to meet global regulatory and manufacturing standards.
SCG Cell Therapy has also recently received support from Singapore’s Economic Development Board (EDB), to expand local research and development, and to expand cell therapy manufacturing capabilities. This would go hand in hand with SCG’s Cell Therapy products, which are being produced at HSA-CTF (Health Sciences Authority-Covid Treatment Facilities).
Currently, SCG has multiple collaborations with local research organisations such as A*STAR, ACTRIS, National University Health System (NUHS), and NationalFCancer Centre Singapore (NCCS), and is looking to secure more collaborations for the sharing of knowledge and to accelerate the development of cell therapy.
Images: Unsplash (featured) and SCG Cell Therapy